
Belay Ascent™
Proprietary Sequencing for
Aneuploidy in CSF
Ascent™ assesses chromosome arm-level and focal losses and gains via low-pass whole genome sequencing (LP-WGS) of tumor-derived nucleic acid in CSF to help inform the diagnosis and management of confirmed or suspected primary and secondary CNS malignancies.
Why Choose Belay Ascent™?
- Pairing Ascent with Summit™ 2.0 can provide critical information for informing the diagnosis and management of CNS cancers using a single CSF specimen.*
- Ascent is highly sensitive, using low amounts of input nucleic acid.
- Ascent employs proprietary technology exclusively licensed for use in CSF by Belay Diagnostics to detect chromosome arm-level losses and gains in confirmed or suspected CNS tumors.

Understanding the Science
Biologic and Technical Basis for Chromosome Arm-Level Alteration Analysis in CNS Malignancies
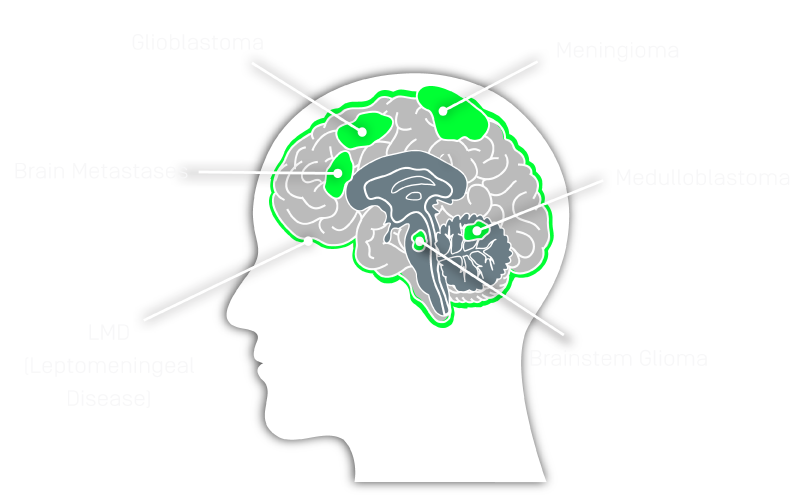
- Chromosome arm-level losses and gains occur in most primary and metastatic CNS cancers1,2
- Metastatic CNS cancers can be detected by the presence of chromosomal alterations across the genome at multiple locations (referred to as “chromosomal instability”)2
- Methods to detect chromosomal alterations in CSF have demonstrated superior sensitivity for detecting CNS cancers compared to the gold-standard of CSF cytology2
- Chromosome arm-level losses and gains can be detected via LP-WGS in CSF using a small quantity of input nucleic acid, making them excellent markers for CNS cancer.
Tumor-Specific Chromosomal Signatures and Clinical Differentiation
- Some primary CNS cancers can be identified by characteristic conserved patterns of arm-level losses or gains:
- Glioblastomas are characterized by Chr. 7p,q gains and Chr. 10 p,q losses1,2
- IDH-mutant oligodendrogliomas canonically exhibit co-deletion of Chr. 1p and Chr. 19q2
- Medulloblastomas are characterized by chromosome arm losses and gains rather than mutations in specific genes4
- Meningiomas are risk-stratified by arm losses3
- Cancer of the breast, lung, and melanoma are most likely to metastasize to the CNS; each has shown chromosome arm-level losses and gains detectable in CSF-tDNA1,2
- GBM and CNS lymphoma can appear similar in imaging but have drastically different patterns of chromosomal loss and gain. Accurate characterization is critical; these cancers have significantly different treatment.2
- Identifying chromosomal alterations can aid in distinguishing neoplasia versus non-neoplastic disease when other tools yield non-diagnostic or negative results2
ASCENTTM DEMONSTRATES EXCEPTIONAL EQUIVALENCE TO STANDARD METHODS
% Concordance of Events: Ascent to CMA/NGS, Tissue (n=48)

ASCENTTM IN CSF IS HIGHLY CONCORDANT TO TISSUE
% Concordance of Events: Ascent in CSF to Tissue Profiling (n=32)


Assay Specifications
| Clinical Performance | Whole arm losses or gains on all chromosomes (only q arms for acrocentric chromosomes 13, 14, 15, 21, and 22) |
| Sample Requirements | ≥ 6 mL of CSF*. A sample of < 6ml of CSF will be processed and results reported provided the sample meets established reporting thresholds |
| Shipping and Transport Temperature | Standard CSF collection tube. Must be shipped in Belay Diagnostics specimen shipping kit within 24 hours of CSF collection and received at Belay Diagnostics within 48 hours. Maintain specimen at room temperature. Do not freeze or refrigerate unless instructed to do so. |
| Methodology | Low-pass whole genome sequencing |
| Orders & Results | Include test requisition in shipping kit or fax form to 800-501-9246. Test results available via fax, encrypted email, or Belay portal. |
| Turnaround Time | Average 7-10 days from receipt of specimen |
References: 1. Douville C, Curtis S, Summers M, Azad TD, et al. Seq-ing the SINEs of central nervous system tumors in cerebrospinal fluid. Cell Rep Med. 2023 Aug 15;4(8):101148. doi: 10.1016/j.xcrm.2023.101148. Epub 2023 Aug 7. PMID:37552989; PMCID: PMC10439243.2. Zheng Y, Ahmad K, Henikoff S. Total whole-arm chromosome losses predict malignancy in human cancer. Proc Natl Acad Sci U S A. 2025 May 6;122(18):e2505385122. doi: 10.1073/pnas.2505385122. Epub 2025 May 2.3. Liu APY, Smith KS, Kumar R, Paul L, et al. Serial assessment of measurable residual disease in medulloblastoma liquid biopsies. Cancer Cell. 2021 Nov 8;39(11):1519-1530.e4. doi: 10.1016/j.ccell.2021.09.012. Epub 2021 Oct 21.4. Sahm F, Aldape KD, Brastianos PK, Brat DJ, et al. cIMPACT-NOW update 8: Clarifications on molecular risk parameters and recommendations for WHO grading of meningiomas. Neuro Oncol. 2025 Feb 10;27(2):319-330. doi: 10.1093/neuonc/noae170.5. Nie Q, Schilter KF, Hernandez KM, Adams JN, et al. Analytical Validation and Clinical Sensitivity of the Belay Summit Assay for the Detection of DNA Variants in Cerebrospinal Fluid of Primary and Metastatic Central Nervous System Cancer. J Mol Diagn. 2025 Jul;27(7):615-629. doi: 10.1016/j.jmoldx.2025.03.010. Epub 2025 Apr 23.
This test was developed, and its performance characteristics determined by Belay Diagnostics, which is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA) as qualified to perform high complexity clinical testing. It has not been cleared or approved by the U.S. Food and Drug Administration (FDA). This test may be used for clinical purposes.
